The relationship between microbiota and renal disease is one of the most interesting arguments of recent years, the microbiota interferes with renal function through its own metabolites, such as trimethylamine (TMA) and short chain fatty acids (SCFA).
Uremic toxin Indole sulfate and microbiota
Indole sulfate is a metabolite of the food tryptophan that acts as a cardiotoxin and a uremic toxin.
In human, high concentrations of this metabolite in the blood are associated with the development and progression of chronic renal disease, being a uremic toxime, stimulates glomerular sclerosis and interstitial renal fibrosis.
Studies suggest that:
Patients with CKD develop higher plasma levels of TMAO and a different composition of the intestinal microbiota than healthy subjects.
Increased TMAO levels in patients with CKD are directly caused by dysbiosis of the microbiota.
TMAO (trimelitalamine-N-oxide) → cardiovascular risk and thrombosis
Indoxylsulphate → renal thrombosis disease progression, cardiovascular disease, anemia
p-cresyl sulfate → renal and vascular disease progression, pro-inflammatory effects
The role of short-chain fatty acids and the microbiota
Microbiota influences renal function and the production of SCFA, short chain fatty acids (SCFA such as acetate, butyrate and propionate) have anti-inflammatory, anti-cancer, antibacterial and antidiabetic effects.
THE ROLE OF INTESTINAL MICROBIOTA AND CHRONIC KIDNEY DISEASE
IN THE HUMAN PATIENT
The intestinal microbiota exerts important metabolic functions that can be modified with the diet, because it uses carbohydrates and proteins present in the intestinal lumen.
Through sucrose fermentation, carbohydrates are converted into short-chain fatty acids.
Through proteolytic fermentation, on the other hand, there is production of phenols, indole, amines and ammonium, potentially toxic metabolites that reduce the circulating levels of SCFA.
A food rich in proteins and fats increases degradation products such as p-cresyl sulfate and indoxyl sulfate compounds almost always increased in patients with CKD and considered markers of disease and uremic toxins.
Short chain fatty acids (SCFA) and dysbiosis
SCFA are produced in the colon and small intestine, are produced after carbohydrate fermentation and have positive effects on the intestinal microbiome and mucosa, have anti-inflammatory and protective effects on immune function and intestinal barrier integrity.
The main compounds are:
Acetic acid
Butyric acid (it is an anti-inflammatory of the intestine, reduces diarrhea and is used to treat inflammatory diseases)
Propionic acid
Valeric acid
Once absorbed and entered the circulation, SCFA have a favourable influence on renal function, are able to counteract a series of pathogenic mechanisms involved in renal damage and have antioxidant, immunoregulatory, anti-inflammatory, antihypertensive and hypoglycemic actions.
When short-chain fatty acids are deficient in the diet
If they are deficient one encounters dysbiosis resulting in the development of diseases such as type 2 diabetes, rheumatoid disease, IBD, MICI, intestinal inflammation, autoimmune diseases, chronic renal failure.
Dysbiosis contributes to the development of kidney diseases, it is clear and indisputable that there is a strong correlation between intestinal inflammation, dysbiosis and progression of chronic renal failure, (so-called gut-renal axis).
The microbiota produces some of the most important uremic toxins
In chronic renal failure the intestinal microbiota is directly involved in the increase of uremic toxins, because it is the dysbiosis induced by the uremia itself that increases the concentrations of toxins in the circulation with a consequent reduction of renal excretion and worsening of the disease.
Faecal microbiome and serum concentrations of indole sulfate are higher in cats with chronic renal disease.
If intestinal dysbiosis has been documented in humans with chronic renal disease (CKD), it is believed to contribute to the production of uremic toxins such as indole sulfate and p-cresol sulfate, even in cats and dogs with chronic renal failure.
In one study faecal samples from 30 cats belonging to stage 2 and stage 4 renal disease clients were analysed to measure serum concentrations of indole sulfate and p-cresol sulfate.
Results:
Cats with CKD had a reduced diversity and richness of faecal bacteria. The concentration of indole sulfate was significantly increased in cats with CKD, and cats with stage 2 CKD had a very similar load of uremic toxins to cats with advanced disease.
How to integrate short-chain fatty acids into the diet of cats and dogs and reduce uremic toxins and indole sulfate
Dietary fibers and fructooligosaccharides (FOS), obtained by hydrolysis of inulin of vegetable origin, are now recognized as “prebiotics”, favorably modify the composition of the intestinal microbiota, stimulate growth and metabolic activity of Bifidobacteri and Lactobacilli to saccharolytic metabolism.
Integration of short-chain fats through:
PREBIOTICS: substance present in food, not absorbed or digested by the body, but metabolized by intestinal bacteria, promotes the development of classes of symbiotic bacteria.
Recent studies show that serum concentrations of PCS and IS are reduced by oral inulin intake. Thanks to the metabolic release of short-chain fatty acids. Inulin reduces nitrogen absorption and also reduces the load of nitrogen waste at the renal level.
- Inulin extracted from chicory root
- Fructoligosaccharides (FOS) promote the growth of bifid and lactobacilli
- Kefir
- Apple pectin binds urea / ammonia
- Modulate dog and cat microbiota to make them produce less uremic toxins
with the administration of

⦁ Symbiotics and Probiotics: at least 9 strains including streptococcus, bifidobacteria and lactobacilli
Probiotics in dog cat renal failure What strains?
Lactobacillus casei Shirota (LcS) —> Reduces serum urea levels by 10% after LcS dietary intervention in CKD 3 and 4 stage patients.
Lactobacillus acidophilus and Bifidobacterium bifidum + Inulin = increases the population of bifidobacteria
Lactobacillus plantarum, Lactobacillus casei subsp. Rhamnosus, Lactobacillus gaseri, Bifidobacterium infantis, Bifidobacterium longum, Lactobacillus acidophilus, Lactobacillus salivarius, Lactobacillus sporogenes, Streptococcus thermophilus, inulin and tapioca starch resistant = Probinul Neutral —-> The symbiotic agent can significantly reduce plasma levels of total p-cresol in patients with stage CKD 3 and 4.
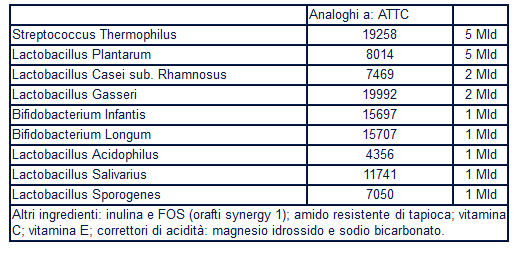
Lactobacillus acidophilus, Lactobacillus casei, Lactobacillus lactis, Bifidobacterium bifidum, Bifidobacterium longum eBifidobacterium infantis —–> Urea levels improved significantly after integration with probiotics.
https://elicats.it/store/petshop/probinul-5-neutro/
Lactobacillus casei, Lactobacillus cidophilus, Lactobacillus bulgarigus, Lactobacillus rhamnosus, Bifido, bacterio breve, Bifidobacterium longum, Streptococcus thermophilus and fructoligosaccharide prebiotico. ——–> Urea nitrogen levels in patients with chronic renal failure have decreased.
Nine strains other than the genera Lactobacillus, Bifidobacterium and Streptococcus. High molecular weight inulin, fructooligosaccharides and galactoligosaccharides and probiotic component. ———> Symbiotic therapy has led to statistically significant and potentially clinically relevant reductions in serum levels of IS and PCS. (Indoxylsulphate – p-cresyl sulfate)
Azodyl for Cats and Dogs: Renal (Kidney) Enteric Dialysis Chronic kidney disease
[irp posts=”13495″ name=”Azodyl in chronic kidney disease CKD cats dogs”]
I dedicate this personal research to a dear friend and her cat suffering from chronic renal failure at stage 3 CKD.
copying is forbidden, you can quote the source 😉
THE CONTENT ON THIS WEBSITE IS TO BE USED FOR INFORMATIONAL PURPOSES ONLY. YOU ACKNOWLEDGE THAT THE CONTENT IS IN NO CASE TO BE USED AS A SUBSTITUTE FOR PROFESSIONAL MEDICAL ADVICE OR ADVICE FROM VETERINARIANS, NOR IS IT INTENDED TO BE RELIED UPON BY ANY PERSON OR ENTITY FOR PURPOSES OF MEDICAL DIAGNOSIS OR TREATMENT . IN NO CASE CAN BE USED AS A PRESCRIPTION OF A TREATMENT OR REPLACE A SPECIALIST EXAMINATION OR THE DIRECT RELATIONSHIP WITH YOUR VETERINARIAN/DOCTOR

